
Atomic electron transition
Change of an electron between energy levels within an atom / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Electronic transition?
Summarize this article for a 10 year old
In atomic physics and chemistry, an atomic electron transition (also called an atomic transition, quantum jump, or quantum leap) is an electron changing from one energy level to another within an atom[1] or artificial atom.[2] The time scale of a quantum jump has not been measured experimentally. However, the Franck–Condon principle binds the upper limit of this parameter to the order of attoseconds.[3]
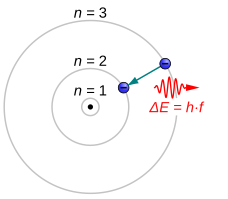
Electrons jumping to energy levels of smaller n emit electromagnetic radiation in the form of a photon. Electrons can also absorb passing photons, which drives a quantum jump to a level of higher n. The larger the energy separation between the electron's initial and final state, the shorter the photons' wavelength.[4]