
Brilaroxazine
Experimental atypical antipsycotic / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Brilaroxazine?
Summarize this article for a 10 year old
Brilaroxazine (developmental code name RP5063), also known as oxaripiprazole,[1][2] is an investigational atypical antipsychotic which is under development by Reviva Pharmaceuticals for the treatment of neuropsychiatric and inflammatory disorders.[3][5][6][7] It has currently completed the first of two phase III clinical trials for schizophrenia. Reviva Pharmaceuticals also intends to investigate brilaroxazine for the treatment of bipolar disorder, major depressive disorder, attention deficit hyperactivity disorder (ADD/ADHD), psychosis/agitation associated with Alzheimer's disease, Parkinson's disease psychosis, as well as the inflammatory disorders pulmonary arterial hypertension (PAH), idiopathic pulmonary fibrosis (IPF), and psoriasis (topical gel).[3][8] The FDA granted brilaroxazine orphan drug designation for the treatment of PAH and IPF.
 | |
| Clinical data | |
|---|---|
| Other names | RP5063; oxaripiprazole [1][2] |
| Routes of administration | By mouth |
| Drug class | Atypical antipsychotic |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >80% [3] |
| Protein binding | >99% |
| Metabolism | Liver (mostly via CYP3A4 (64%) and CYP2D6 (17%)) [4] |
| Elimination half-life | 55 hours [3] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
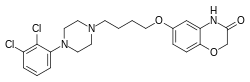
| Formula | C22H25Cl2N3O3 |
| Molar mass | 450.36 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Brilaroxazine is a third-generation antipsychotic and dopamine-serotonin system modulator due to its unique actions on dopamine and serotonin neurotransmitter systems compared to other antipsychotics.[9][10][11] Clinical data from phase I, phase II (NCT01490086), and phase III (NCT05184335) trials suggest that brilaroxazine may have favorable efficacy and a significantly improved side effect profile compared to existing third-generation drugs.[9][12][13]