
Citrulline
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Citrulline?
Summarize this article for a 10 year old
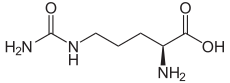
The organic compound citrulline is an α-amino acid.[2] Its name is derived from citrullus, the Latin word for watermelon. Although named and described by gastroenterologists since the late 19th century, it was first isolated from watermelon in 1914 by Japanese researchers Yotaro Koga and Ryo Odake[3][note 1] and further codified by Mitsunori Wada of Tokyo Imperial University in 1930.[4] It has the formula H2NC(O)NH(CH2)3CH(NH2)CO2H. It is a key intermediate in the urea cycle, the pathway by which mammals excrete ammonia by converting it into urea. Citrulline is also produced as a byproduct of the enzymatic production of nitric oxide from the amino acid arginine, catalyzed by nitric oxide synthase.[5]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-Amino-5-(carbamoylamino)pentanoic acid[1] | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3DMet | |
| 1725417, 1725415 D, 1725416 L | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank |
|
| ECHA InfoCard | 100.006.145 |
| EC Number |
|
| 774677 L | |
| KEGG |
|
| MeSH | Citrulline |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H13N3O3 | |
| Molar mass | 175.188 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| log P | −1.373 |
| Acidity (pKa) | 2.508 |
| Basicity (pKb) | 11.489 |
| Thermochemistry | |
Heat capacity (C) |
232.80 J K−1 mol−1 |
Std molar entropy (S⦵298) |
254.4 J K−1 mol−1 |
| Related compounds | |
Related alkanoic acids |
|
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |