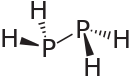Diphosphane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Diphosphane?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
This article is about P2H4. For coordination chemistry of its substituted derivatives, see diphosphines.
Diphosphane, or diphosphine, is an inorganic compound with the chemical formula P2H4. This colourless liquid is one of several binary phosphorus hydrides. It is the impurity that typically causes samples of phosphine to ignite in air.
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Diphosphane | |||
| Systematic IUPAC name
Diphosphane (substitutive) Tetrahydridodiphosphorus(P—P) (additive) | |||
| Other names
Diphosphine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| H4P2 | |||
| Molar mass | 65.980 g·mol−1 | ||
| Melting point | −99 °C (−146 °F; 174 K) | ||
| Boiling point | 63.5 °C (146.3 °F; 336.6 K) (Extrapolated, decomposes) | ||
| Related compounds | |||
Other anions |
ammonia hydrazine triazane | ||
Other cations |
diphosphines | ||
Related Binary Phosphorus halides |
diphosphorus tetrafluoride diphosphorus tetrachloride diphosphorus tetrabromide diphosphorus tetraiodide | ||
Related compounds |
phosphane triphosphane diphosphene diphosphenes | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close