
Mercuric amidochloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Mercuric amidochloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Millon's base" redirects here. For the analytical reagent used to detect the presence of soluble proteins, see Millon's reagent.
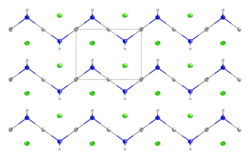
Mercuric amidochloride is an inorganic compound with the formula Hg(NH2)Cl.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Mercuric azanide chloride | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.030.292 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Hg(NH2)Cl | |
| Molar mass | 252.065 g/mol |
| Appearance | White crystalline solid or white amorphous powder[1] |
| Odor | Odorless[1] |
| Density | 5.56 g/cm3 |
| Boiling point | Sublimes[1] |
| 0.14 g in 100 mL of cold water 100 g in 100 mL of hot water (decomposes)[1] | |
| Solubility | Soluble in warm hydrochloric acid, nitric acid and acetic acid, insoluble in ethanol[1] |
| Pharmacology | |
| D08AK01 (WHO) | |
| Hazards | |
| GHS labelling:[1] | |
   | |
| Danger | |
| H300, H310, H330, H373, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+P316, P302+P352, P304+P340, P316, P319, P320, P321, P330, P361+P364, P391, P403+P233, P405, P501 | |
| Flash point | Non-combustible |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
|
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close