
Sodium carbonate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sodium carbonate?
Summarize this article for a 10 year old
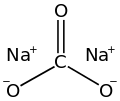
Sodium carbonate (also known as washing soda, soda ash and soda crystals) is the inorganic compound with the formula Na2CO3 and its various hydrates. All forms are white, odourless, water-soluble salts that yield alkaline solutions in water. Historically, it was extracted from the ashes of plants grown in sodium-rich soils, and because the ashes of these sodium-rich plants were noticeably different from ashes of wood (once used to produce potash), sodium carbonate became known as "soda ash".[12] It is produced in large quantities from sodium chloride and limestone by the Solvay process, as well as by carbonating sodium hydroxide which is made using the Chlor-alkali process.
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Sodium carbonate | |
| Other names
Soda ash, washing soda, soda crystals, sodium trioxocarbonate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.007.127 |
| EC Number |
|
| E number | E500(i) (acidity regulators, ...) |
PubChem CID |
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Na2CO3 | |
| Molar mass | 105.9888 g/mol (anhydrous) 286.1416 g/mol (decahydrate) |
| Appearance | White solid, hygroscopic |
| Odor | Odorless |
| Density | |
| Melting point | 851 °C (1,564 °F; 1,124 K) (Anhydrous) 100 °C (212 °F; 373 K) decomposes (monohydrate) 33.5 °C (92.3 °F; 306.6 K) decomposes (heptahydrate) 34 °C (93 °F; 307 K) (decahydrate)[2][3] |
Anhydrous, g/100 mL:
| |
| Solubility | Soluble in aq. alkalis,[4] glycerol Slightly soluble in aq. alcohol Insoluble in CS2, acetone, alkyl acetates, alcohol, benzonitrile, liquid ammonia[5] |
| Solubility in glycerine | 98.3 g/100 g (155 °C)[5] |
| Solubility in ethanediol | 3.46 g/100 g (20 °C)[6] |
| Solubility in dimethylformamide | 0.5 g/kg[6] |
| Acidity (pKa) | 10.33 |
| −4.1·10−5 cm3/mol[2] | |
Refractive index (nD) |
1.485 (anhydrous) 1.420 (monohydrate)[3] 1.405 (decahydrate) |
| Viscosity | 3.4 cP (887 °C)[6] |
| Structure | |
| Monoclinic (γ-form, β-form, δ-form, anhydrous)[7] Orthorhombic (monohydrate, heptahydrate)[1][8] | |
| C2/m, No. 12 (γ-form, anhydrous, 170 K) C2/m, No. 12 (β-form, anhydrous, 628 K) P21/n, No. 14 (δ-form, anhydrous, 110 K)[7] Pca21, No. 29 (monohydrate)[1] Pbca, No. 61 (heptahydrate)[8] | |
| 2/m (γ-form, β-form, δ-form, anhydrous)[7] mm2 (monohydrate)[1] 2/m 2/m 2/m (heptahydrate)[8] | |
α = 90°, β = 101.35(8)°, γ = 90° | |
| Octahedral (Na+, anhydrous) | |
| Thermochemistry | |
Heat capacity (C) |
112.3 J/mol·K[2] |
Std molar entropy (S⦵298) |
135 J/mol·K[2] |
Std enthalpy of formation (ΔfH⦵298) |
−1130.7 kJ/mol[2][6] |
Gibbs free energy (ΔfG⦵) |
−1044.4 kJ/mol[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Irritant |
| GHS labelling: | |
 [9] [9] | |
| Warning | |
| H319[9] | |
| P305+P351+P338[9] | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
4090 mg/kg (rat, oral)[11] |
| Safety data sheet (SDS) | MSDS |
| Related compounds | |
Other anions |
Sodium bicarbonate |
Other cations |
Lithium carbonate Potassium carbonate Rubidium carbonate Cesium carbonate |
Related compounds |
Sodium sesquicarbonate Sodium percarbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
