
Solriamfetol
Medication used for the treatment of excessive sleepiness / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Solriamfetol?
Summarize this article for a 10 year old
Solriamfetol, sold under the brand name Sunosi, is a wakefulness-promoting medication used in the treatment of excessive sleepiness related to narcolepsy and sleep apnea.[3][5][6] It is taken by mouth.[3]
 | |
| Clinical data | |
|---|---|
| Trade names | Sunosi |
| Other names | SKL-N05, ADX-N05, ARL-N05, YKP10A, R228060, and JZP-110; (R)-2-amino-3-phenylpropylcarbamate hydrochloride |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619040 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Norepinephrine–dopamine reuptake inhibitors |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~95%[3] |
| Protein binding | 13.3–19.4%[3] |
| Metabolism | Minimal[3] |
| Elimination half-life | ~7.1 hours[3] |
| Excretion | Urine (95% unchanged) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
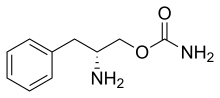
| Formula | C10H14N2O2 |
| Molar mass | 194.234 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Common side effects of solriamfetol include headache, nausea, anxiety, and trouble sleeping.[3] It is a norepinephrine–dopamine reuptake inhibitor (NDRI) and is thought to work by increasing levels of the neurotransmitters norepinephrine and dopamine in the brain.[3][5]
The drug was discovered by a subsidiary of SK Group, which licensed rights outside of eleven countries in Asia to Aerial Pharma in 2011.[7] In addition to its approved indication of excessive sleepiness, solriamfetol is under development for certain other uses including the treatment of attention deficit hyperactivity disorder (ADHD), binge eating disorder, and circadian rhythm sleep disorders.[8]