
Trimethylaluminium
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trimethyl aluminium?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Trimethylaluminium is one of the simplest examples of an organoaluminium compound. Despite its name it has the formula Al2(CH3)6 (abbreviated as Al2Me6 or TMA), as it exists as a dimer. This colorless liquid is pyrophoric. It is an industrially important compound, closely related to triethylaluminium.[3][4]
Quick Facts Names, Identifiers ...
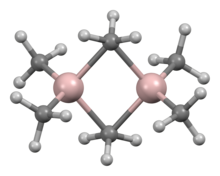 | |
 | |
| Names | |
|---|---|
| IUPAC name
Trimethylalumane | |
| Other names
Trimethylaluminum; aluminium trimethyl; aluminum trimethyl | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.000.776 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C6H18Al2 | |
| Molar mass | 144.17 g/mol 72.09 g/mol (C3H9Al) |
| Appearance | Colorless liquid |
| Density | 0.752 g/cm3 |
| Melting point | 15 °C (59 °F; 288 K) |
| Boiling point | 125–130 °C (257–266 °F; 398–403 K)[1][2] |
| Reacts | |
| Vapor pressure |
|
| Viscosity |
|
| Thermochemistry | |
Heat capacity (C) |
155.6 J/mol·K[2] |
Std molar entropy (S⦵298) |
209.4 J/mol·K[2] |
Std enthalpy of formation (ΔfH⦵298) |
−136.4 kJ/mol[2] |
Gibbs free energy (ΔfG⦵) |
−9.9 kJ/mol[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Pyrophoric |
| GHS labelling: | |
  [1] [1] | |
| Danger | |
| H250, H260, H314[1] | |
| P222, P223, P231+P232, P280, P370+P378, P422[1] | |
| NFPA 704 (fire diamond) | |
| Flash point | −17.0 °C (1.4 °F; 256.1 K)[1] |
| Related compounds | |
Related compounds |
Triethylaluminium |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
