
Beryllium carbonate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Beryllium carbonate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Beryllium carbonate is a chemical compound with the chemical formula BeCO3.
Quick Facts Identifiers, Properties ...
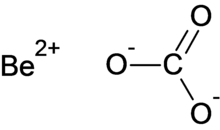 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.032.740 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UN number | 1566 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| BeCO3 | |
| Molar mass | 69.020 g·mol−1 |
| Melting point | 54 °C (129 °F; 327 K) |
| Boiling point | 100 °C (212 °F; 373 K) decomposes |
| 0.36 g/100 mL | |
| Thermochemistry | |
Heat capacity (C) |
65 J/mol·K[1] |
Std molar entropy (S⦵298) |
52 J/mol·K[1] |
Std enthalpy of formation (ΔfH⦵298) |
-1025 kJ/mol[1] |
Gibbs free energy (ΔfG⦵) |
-948 kJ/mol[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Toxic (T) Irritant (Xi) |
| GHS labelling:[2] | |
   | |
| Danger | |
| H301, H315, H317, H319, H330, H335, H350i, H372, H411 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
150 mg/kg (guinea pig) |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[3] |
REL (Recommended) |
Ca C 0.0005 mg/m3 (as Be)[3] |
IDLH (Immediate danger) |
Ca [4 mg/m3 (as Be)][3] |
| Related compounds | |
Other cations |
Magnesium carbonate Calcium carbonate Strontium carbonate Barium carbonate Radium carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
