
Ethylenediaminetetraacetic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ethylenediaminetetraacetic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
This article is about the chemical. For the medication, see Sodium calcium edetate.
"EDTA" redirects here. For other uses, see EDTA (disambiguation).
"Versene" redirects here. Not to be confused with Versine.
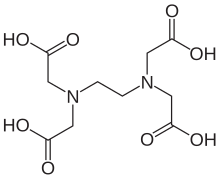
Ethylenediaminetetraacetic acid (EDTA), also called EDTA acid after its own abbreviation, is an aminopolycarboxylic acid with the formula [CH2N(CH2CO2H)2]2. This white, water-insoluble solid is widely used to bind to iron (Fe2+/Fe3+) and calcium ions (Ca2+), forming water-soluble complexes even at neutral pH. It is thus used to dissolve Fe- and Ca-containing scale as well as to deliver iron ions under conditions where its oxides are insoluble. EDTA is available as several salts, notably disodium EDTA, sodium calcium edetate, and tetrasodium EDTA, but these all function similarly.[4]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
N,N′-(Ethane-1,2-diyl)bis[N-(carboxymethyl)glycine][1] | |
| Systematic IUPAC name
2,2′,2′′,2′′′-(Ethane-1,2-diyldinitrilo)tetraacetic acid[1] | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | EDTA, H4EDTA |
| 1716295 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.000.409 |
| EC Number |
|
| 144943 | |
| KEGG |
|
| MeSH | Edetic+Acid |
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| UN number | 3077 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H16N2O8 | |
| Molar mass | 292.244 g·mol−1 |
| Appearance | Colourless crystals |
| Density | 0.860 g cm−3 (at 20 °C) |
| log P | −0.836 |
| Acidity (pKa) | 2.0, 2.7, 6.16, 10.26[2] |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−1765.4 to −1758.0 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−4461.7 to −4454.5 kJ mol−1 |
| Pharmacology | |
| S01XA05 (WHO) V03AB03 (WHO) (salt) | |
| |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H319 | |
| P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1000 mg/kg (oral, rat)[3] |
| Related compounds | |
Related alkanoic acids |
|
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
