
Benzbromarone
Chemical compound / From Wikipedia, the free encyclopedia
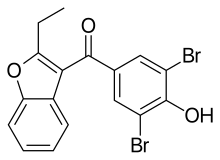
Benzbromarone is a uricosuric agent and non-competitive inhibitor of xanthine oxidase[1] used in the treatment of gout, especially when allopurinol, a first-line treatment, fails or produces intolerable adverse effects. It is structurally related to the antiarrhythmic amiodarone.[2]
Quick Facts Clinical data, AHFS/Drugs.com ...
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.020.573 |
| Chemical and physical data | |
| Formula | C17H12Br2O3 |
| Molar mass | 424.088 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 161 to 163 °C (322 to 325 °F) |
| |
| |
| | |
Close
Benzbromarone is highly effective and well tolerated,[3][4][5][6] and clinical trials as early as 1981 and as recently as April 2008 have suggested it is superior to both allopurinol, a non-uricosuric xanthine oxidase inhibitor, and probenecid, another uricosuric drug.[7][8]