
Meloxicam
Nonsteroidal anti-inflammatory drug (NSAID) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Meloxicam?
Summarize this article for a 10 year old
Meloxicam, sold under the brand name Mobic among others, is a nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammation in rheumatic diseases and osteoarthritis.[7][8] It is used by mouth or by injection into a vein.[8][9] It is recommended that it be used for as short a period as possible and at a low dose.[8]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Mobic, Metacam, Anjeso, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601242 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 89%[6] |
| Protein binding | 99.4%[6] |
| Metabolism | Liver (CYP2C9 and 3A4-mediated)[6] |
| Elimination half-life | 20 hours[6] |
| Excretion | Urine and feces equally[6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.113.257 |
| Chemical and physical data | |
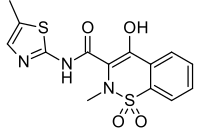
| Formula | C14H13N3O4S2 |
| Molar mass | 351.40 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include abdominal pain, dizziness, swelling, headache, and a rash.[8] Serious side effects may include heart disease, stroke, kidney problems, and stomach ulcers.[8] Use is not recommended in the third trimester of pregnancy.[8] It blocks cyclooxygenase-2 (COX-2) more than it blocks cyclooxygenase-1 (COX-1).[8] It is in the oxicam family of chemicals and is closely related to piroxicam.[8]
Meloxicam was patented in 1977 and approved for medical use in the United States in 2000.[8][10] It was developed by Boehringer Ingelheim; however, it is also available as a generic medication.[8] In 2021, it was the 32nd most commonly prescribed medication in the United States, with more than 18 million prescriptions.[11][12] An intravenous version of meloxicam (Anjeso) was approved for medical use in the United States in February 2020.[13][9]