
Methyl fluoroacetate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Methyl fluoroacetate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Methyl fluoroacetate (MFA) is an organic compound with the chemical formula FCH2CO2CH3. It is an extremely toxic methyl ester of fluoroacetic acid. It is a colorless, odorless liquid at room temperature. It is used as a laboratory chemical and as a rodenticide. Because of its extreme toxicity, MFA was studied for potential use as a chemical weapon.[1]
Quick Facts Names, Identifiers ...
 | |
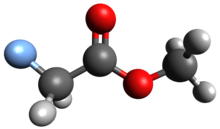 | |
| Names | |
|---|---|
| Preferred IUPAC name
Methyl fluoroacetate | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.563 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| FCH2CO2CH3 | |
| Molar mass | 92.069 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Odorless or faint fruity |
| Melting point | −40 °C (−40 °F; 233 K) |
| Boiling point | 104 °C (219 °F; 377 K) |
| 117 g/L at 25 °C | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Extremely toxic |
| GHS labelling: | |
    | |
| Danger | |
| H226, H300, H315, H319, H335, H400 | |
| P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
| Flash point | −32 °C (−26 °F; 241 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
6 mg/kg (mice) |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
The general population is not likely to be exposed to methyl fluoroacetate. People who use MFA for work, however, can breathe in or have direct skin contact with the substance.[2]