
Perfluorohexane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Perfluorohexane?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
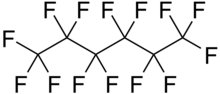
Perfluorohexane (C6F14), or tetradecafluorohexane, is a fluorocarbon. It is a derivative of hexane in which all the hydrogen atoms are replaced by fluorine atoms. It is used in one formulation of the electronic cooling liquid/insulator Fluorinert for low-temperature applications due to its low boiling point of 56 °C and freezing point of −90 °C. It is odorless and colorless. Unlike typical hydrocarbons, the structure features a helical carbon backbone.[1] In medical imaging it is used as a contrast agent.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Tetradecafluorohexane | |
| Other names
FC-72, Fluorinert FC-72, Flutec PP1, Perfluoro-compound FC-72 | |
| Identifiers | |
| |
3D model (JSmol) |
|
| Abbreviations | PFH |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.005.987 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6F14 | |
| Molar mass | 338.041845 |
| Appearance | Clear, colorless |
| Odor | Odorless |
| Density | 1.680 kg/m3 (Liquid) |
| Melting point | −90 °C (−130 °F; 183 K) |
| Boiling point | 56 °C (133 °F; 329 K) |
| Vapor pressure | 30.9 kPa (25 °C) |
| Thermal conductivity | 0.057 W/(m·K) |
| Viscosity | 0.64 cP |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
> 5 g/kg (rat, oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
