
Rimonabant
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Rimonabant?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
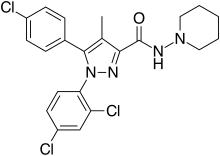
Rimonabant (also known as SR141716; trade names Acomplia, Zimulti)[3] is an anorectic antiobesity drug approved in Europe in 2006 but was withdrawn worldwide in 2008 due to serious psychiatric side effects; it was never approved in the United States.[1][2] Rimonabant is an inverse agonist for the cannabinoid receptor CB1 and was first-in-class for clinical development.[4][5]
Quick Facts Clinical data, AHFS/Drugs.com ...
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Undetermined |
| Protein binding | Nearly 100% |
| Metabolism | Hepatic, CYP3A4 involved |
| Elimination half-life | Variable: 6 to 9 days with normal BMI 16 days if BMI >30 |
| Excretion | Fecal (86%) and renal (3%) |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.978 |
| Chemical and physical data | |
| Formula | C22H21Cl3N4O |
| Molar mass | 463.79 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Close