
Squalene
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Squalene?
Summarize this article for a 10 year old
Squalene is an organic compound. It is a triterpene with the formula C30H50. It is a colourless oil, although impure samples appear yellow. It was originally obtained from shark liver oil (hence its name, as Squalus is a genus of sharks). An estimated 12% of bodily squalene in humans is found in sebum.[5] Squalene has a role in topical skin lubrication and protection.[6]
 | |
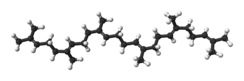 | |
| Names | |
|---|---|
| Preferred IUPAC name
(6E,10E,14E,18E)-2,6,10,15,19,23-Hexamethyltetracosa-2,6,10,14,18,22-hexaene[1] | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3DMet | |
| 1728919 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.003.479 |
| EC Number |
|
| KEGG |
|
| MeSH | Squalene |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C30H50 | |
| Molar mass | 410.730 g·mol−1 |
| Appearance | Colourless oil |
| Density | 0.858 g·cm−3 |
| Melting point | −5 °C (23 °F; 268 K)[2] |
| Boiling point | 285 °C (545 °F; 558 K) at 3.3 kPa[3] |
| log P | 12.188 |
Refractive index (nD) |
1.4956 (at 20 °C) [4] |
| Viscosity | 12 cP (at 20 °C) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 110 °C (230 °F; 383 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Most plants, fungi, and animals produce squalene as biochemical precursor in sterol biosynthesis, including cholesterol and steroid hormones in the human body.[7][8][9] It is also an intermediate in the biosynthesis of hopanoids in many bacteria.[10]
Squalene is an important ingredient in some vaccine adjuvants: The Novartis and GlaxoSmithKline adjuvants are called MF59 and AS03, respectively.[11]
