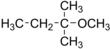
tert-Amyl methyl ether
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tert-Amyl methyl ether?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
tert-Amyl methyl ether (TAME) is an ether used as a fuel oxygenate. TAME derives from C5 distillation fractions of naphtha.[4] It has an ethereous odor.[1] Unlike most ethers, it does not require a stabilizer as it does not form peroxides on storage.[5]
Not to be confused with tert-Amyl ethyl ether.
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methoxy-2-methylbutane | |||
| Other names
tertiary-Amyl methyl ether; TAME; Methoxypentane | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| Abbreviations | TAME | ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.012.374 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C6H14O | |||
| Molar mass | 102.177 g·mol−1 | ||
| Appearance | Clear, colorless liquid | ||
| Density | 0.76-0.78 g/mL[3] | ||
| Melting point | −80 °C (−112 °F; 193 K) | ||
| Boiling point | 86.3 °C (187.3 °F; 359.4 K) | ||
| 10.71 g/L at 20 °C | |||
Refractive index (nD) |
1.3896 | ||
| Hazards | |||
| Flash point | −11 °C (12 °F; 262 K) | ||
| 430 °C (806 °F; 703 K) | |||
| Explosive limits | 1.0-7.1% | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Other names:[6]
- 2-Methoxy-2-methylbutane
- Butane, 2-methoxy-2-methyl-
- 1,1-Dimethylpropyl methyl ether
- Methyl tert-pentyl ether
- Methyl tert-Amyl ether
- 2-Methyl-2-methoxybutane
- Methyl 2-methyl-2-butyl ether
- tert-Pentyl methyl ether
- Tertiary amyl methyl ether
- Methyl 1,1-dimethylpropyl ether
- 2-Methoxy-2-methylbutane