Tributyl phosphate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tributyl phosphate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
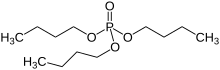
Tributyl phosphate, known commonly as TBP, is an organophosphorus compound with the chemical formula (CH3CH2CH2CH2O)3PO. This colourless, odorless liquid finds some applications as an extractant and a plasticizer. It is an ester of phosphoric acid with n-butanol.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Tributyl phosphate | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.004.365 | ||
| KEGG |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C12H27O4P | |||
| Molar mass | 266.318 g·mol−1 | ||
| Appearance | Colorless to pale-yellow liquid[1] | ||
| Density | 0.9727 g/mL | ||
| Melting point | −80 °C (−112 °F; 193 K) | ||
| Boiling point | 289 °C (552 °F; 562 K) | ||
| 0.4 g/L[2] | |||
| Vapor pressure | 0.004 mmHg (25°C)[1] | ||
Refractive index (nD) |
1.4231 (at 20 °C) [3] | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 146.1 °C (295.0 °F; 419.2 K) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
1189 mg/kg (mouse, oral) 3000 mg/kg (rat, oral)[4] | ||
LC50 (median concentration) |
227 ppm (cat, 4–5 h) 123 ppm (rat, 6 h) 117 ppm (rat) 2529 ppm (rat, 1 h)[4] | ||
LCLo (lowest published) |
2214 ppm (cat, 5 h)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 5 mg/m3[1] | ||
REL (Recommended) |
TWA 0.2 ppm (2.5 mg/m3)[1] | ||
IDLH (Immediate danger) |
30 ppm[1] | ||
| Safety data sheet (SDS) | External MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close