
Bicarbonate buffer system
Buffer system that maintains pH balance in humans / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Bicarbonate buffer system?
Summarize this article for a 10 year old
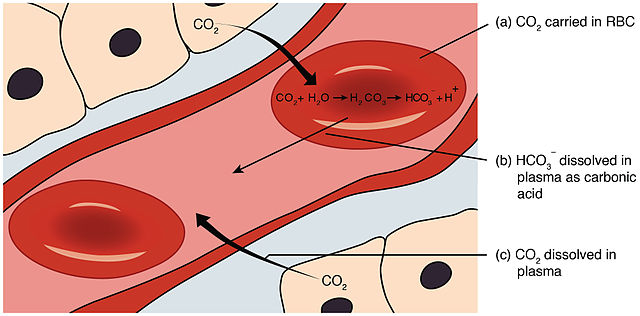
The bicarbonate buffer system is an acid-base homeostatic mechanism involving the balance of carbonic acid (H2CO3), bicarbonate ion (HCO−
3), and carbon dioxide (CO2) in order to maintain pH in the blood and duodenum, among other tissues, to support proper metabolic function.[1] Catalyzed by carbonic anhydrase, carbon dioxide (CO2) reacts with water (H2O) to form carbonic acid (H2CO3), which in turn rapidly dissociates to form a bicarbonate ion (HCO−
3 ) and a hydrogen ion (H+) as shown in the following reaction:[2][3][4]

As with any buffer system, the pH is balanced by the presence of both a weak acid (for example, H2CO3) and its conjugate base (for example, HCO−
3) so that any excess acid or base introduced to the system is neutralized.
Failure of this system to function properly results in acid-base imbalance, such as acidemia (pH < 7.35) and alkalemia (pH > 7.45) in the blood.[5]