
Carbonic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Carbonic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with carbolic acid or carboxylic acid.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Carbonic acid[1] | |
Other names
| |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank | |
| ECHA InfoCard | 100.133.015 |
| EC Number |
|
| 25554 | |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
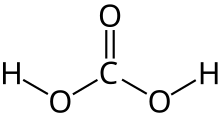
| H 2CO 3 | |
| Appearance | Colorless gas |
| Melting point | −53 °C (−63 °F; 220 K)[2] (sublimes) |
| Boiling point | 127 °C (261 °F; 400 K) (decomposes) |
| Reacts to form carbon dioxide and water | |
| Acidity (pKa) | pKa1 = 3.6 at 25 °C pKa2 = 10.329 |
| Conjugate base | Bicarbonate, carbonate |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Structure | |
| monoclinic | |
| p21/c, No. 14 | |
| - | |
a = 5.392 Å, b = 6.661 Å, c = 5.690 Å (D 2CO 3 at 1.85 GPa, 298 K) | |
Lattice volume (V) |
204.12 Å3 |
Formula units (Z) |
4 formula per cell |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Carbonic acid is a chemical compound with the chemical formula H2CO3. The molecule rapidly converts to water and carbon dioxide in the presence of water. However, in the absence of water, it is (contrary to popular belief) quite stable at room temperature.[4][5] The interconversion of carbon dioxide and carbonic acid is related to the breathing cycle of animals and the acidification of natural waters.[3]
In biochemistry and physiology, the name "carbonic acid" is sometimes applied to aqueous solutions of carbon dioxide. These chemical species play an important role in the bicarbonate buffer system, used to maintain acid–base homeostasis.[6]
