
Gallium trichloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Gallium trichloride?
Summarize this article for a 10 year old
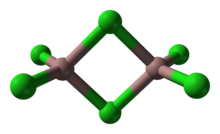
Gallium trichloride is the chemical compound with the formula GaCl3. Solid gallium trichloride exists as a dimer with the formula Ga2Cl6.[1] It is colourless and soluble in virtually all solvents, even alkanes, which is truly unusual for a metal halide. It is the main precursor to most derivatives of gallium and a reagent in organic synthesis.[2]
 | |
 | |
| Names | |
|---|---|
| Other names
Gallium(III) chloride, Trichlorogallium, Trichlorogallane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.033.268 |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| GaCl 3 | |
| Molar mass | 176.073 g/mol |
| Appearance | colorless crystals deliquescent |
| Density | 2.47 g/cm3 2.053 g/cm3 at melting point |
| Melting point | 77.9 °C (172.2 °F; 351.0 K) (anhydrous) 44.4 °C (hydrate) |
| Boiling point | 201 °C (394 °F; 474 K) |
| very soluble | |
| Solubility | soluble in benzene, CCl4, CS2 |
| −63.0·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
4700 mg/kg (rat, oral) |
| Related compounds | |
Other anions |
Gallium(III) fluoride Gallium(III) bromide Gallium(III) iodide |
Other cations |
Aluminium chloride Indium(III) chloride Thallium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
As a Lewis acid, GaCl3 is milder than aluminium trichloride. Gallium(III) is easier to reduce than Al(III), so the chemistry of reduced gallium compounds is more extensive than for aluminium. Ga2Cl4 is known whereas the corresponding Al2Cl4 is not. The coordination chemistry of Ga(III) and Fe(III) are similar, and gallium(III) compounds have been used as diamagnetic analogues of ferric compounds.
